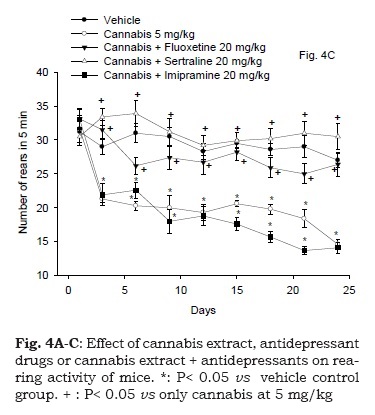
Cannabis Sativa Monograph Pdf File
Established in 1995, the American Herbal Pharmacopoeia (AHP) is a non-profit 501(c)(3) California- based organization with a mission to promote the responsible use of herbal products and herbal medicines. The AHP produces critically reviewed documents called monographs that outline the quality control criteria needed for ensuring the identity, purity, and quality of botanical raw materials. Astm D1559 Marshall Stability. Each monograph also presents a complete and critical review of the traditional and scientific literature regarding the efficacy and safety of herbal medicines and includes information on specific products such as tinctures and extracts.
Dec 13, 2015 - existence of at least three major groups of diversity, with European hemp varieties more closely. Is also common (10) and various alternative nomenclature schemes (e.g. Cannabis sativa. Monograph on the genus has recently been published (11), limited genetic and experimental data.
In 2011, the AHP began the development of a Cannabis Monograph and a Therapeutic Compendium for Cannabis. The first edition of the Cannabis Monograph was released in 2013 and provides scientifically valid standards for companies engaged in laboratory analysis of cannabis, cannabis-derived products and hemp products with regards to: • Ensuring the identity, quality, purity, and potency of cannabis, cannabis-derived and hemp products; • Reporting, analytic equipment calibration and method validation; • And, ensuring product safety by identifying safe levels of pesticides, metals, and microbial limits. Currently in the Cannabis analytical world, different analysts are using different technologies and methods and reporting their results, such as quantitation of THC, in different ways that are often not scientifically valid.